



About Recoupedrol
Recoupedrol is high in glycoside content and backed with 4 human clinical, Toxicological and preclinical studies. The manufacturing process for Recoupedrol uses cane ethanol and water only. Additionally, it is soluble in water. Recoupedrol is also Human safety assessed for one year
Safety of Recoupedrol has been evaluated in 4 different toxicological studies as per OECD guidelines
A. Acute toxicity in rats-LD50> 2000 mg/kg
B. 90 day repeated dose oral toxicity in rats-No Observed Adverse Effect Level (NOAEL) at 1000 mg/kg in male rats & 500 mg/kg in female rats.
C. In-vitro bacterial reverse mutation assay (AMES Test)-
a. Maximum concentration = 5000 mcg/plate
b. Recoupedrol is not mutagenic
D. Pre-natal developmental toxicity study- NOAEL at 1000 mg/kg
Effect of Recoupedrol on patients with Rheumatoid Arthritis
Recoupedrol was tested on 130 patients with Rheumatoid Arthritis in a double-blind placebo controlled study for 3 months and followed by 9 months open label study.
ACR20 and ACR 50
The American College of Rheumatology (ACR) Criteria measures improvement in arthritic symptoms such as pain, swelling, tenderness and stiffness.
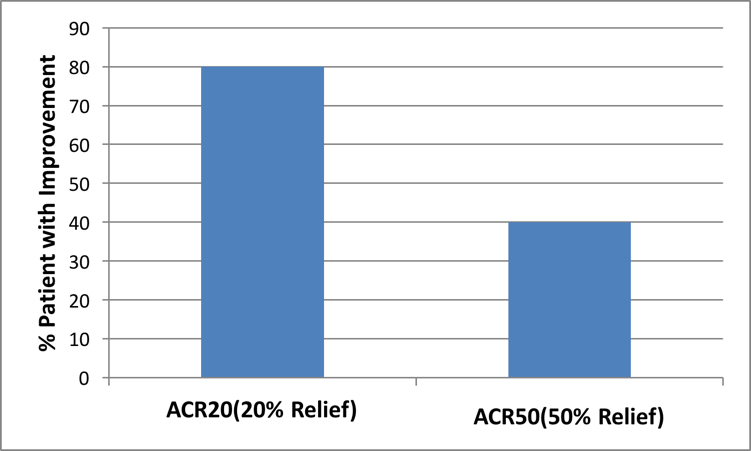
After 12 months of treatment, 80% of the patients showed a 20% improvement (ACR20) in ACR factors like pain, tenderness, swelling and stiffness; 40% percent of the patients showed 50% improvement in the same factors.
Rheumatoid Factor
Rheumatoid Factor (RF) is an auto antibody that is directed against its own tissues (for example- inflammatory tissues). A higher level of RF shows greater probability of destruction of joints. RF, in combination with signs and symptoms can play a role in inflammatory disease prognosis.
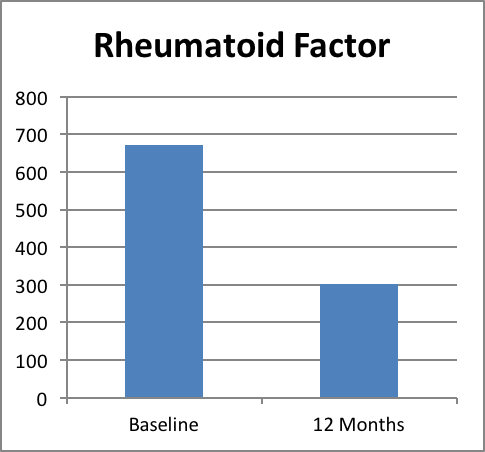
After 12 months of treatment, the RF titre reduced significantly by 55% in patients treated with Recoupedrol
C – reactive protein
C-reactive protein is a substance produced by the liver in response to inflammation.
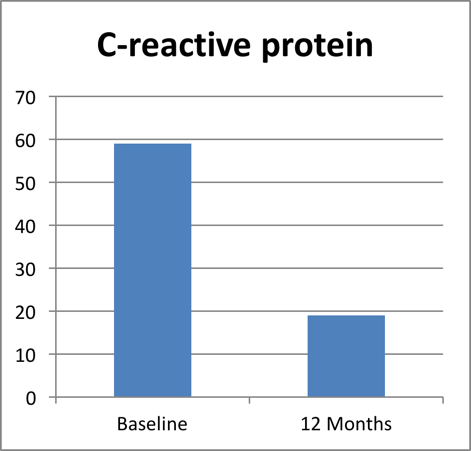
After 12 months of treatment the C-reactive protein reduced significantly by 68 % in patients treated with Recoupedrol
Effect of Recoupedrol on arthritic symptoms (including pain) after 12 months treatment
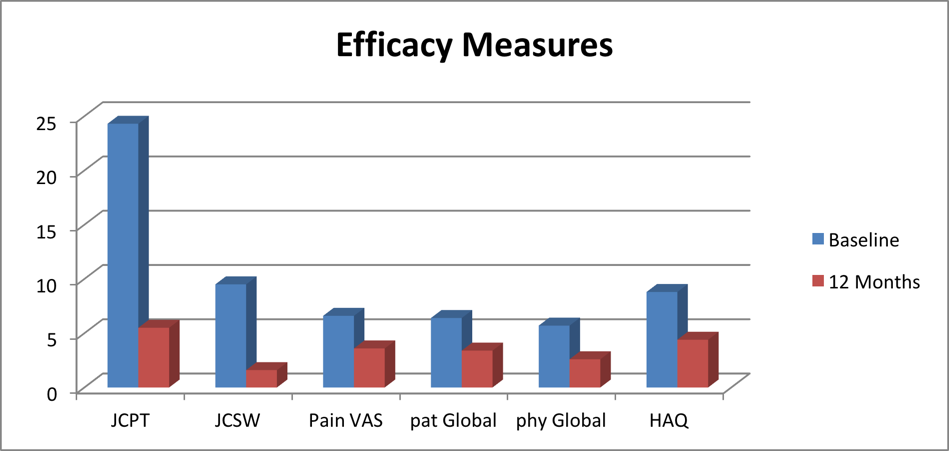
Recoupedrol showed significant improvement in all Arthritic symptoms after 12 months treatment
Chronic inflammatory mediators inhibitory effect of Recoupedrol on in In-vitro Peripheral blood mononuclear cells(PBMC)
Biological response to the inflammatory stimuli leads to continuous release of pro inflammatory mediators like interleukins such as IL-1, IL-6 and TNF-α. These mediators change the proportion of osteoclast (bone resorption) and osteoblast (bone formation). Balanced proportion of these cells is critical for maintenance and repair of the bone. Imbalance of these cells results in chronic inflammation
A continuous release of pro-inflammatory cytokines and interleukins during chronic inflammation disturbs this balance. To balance, we need to inhibit the mediators.
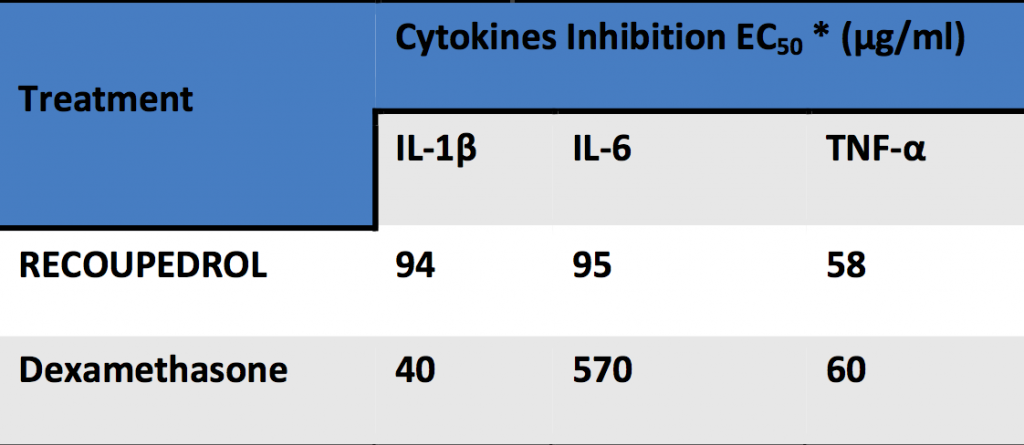
*EC50 – the concentration of a drug that gives half-maximal effective response. Lower the EC50, higher will be the potency of compound Dexamethasone was a positive control
Other Applications of Recoupedrol
Suitable ingredient for health products in the form of capsules, tablets, beverages, cosmetics, energy bars etc.
- POST WORKOUT RECOVERY
- ANTI AGING











